The Metal Reactivity Power Rankings
Highly active metals like potassium sit at the top, while stable elements like gold remain at the bottom, with hydrogen serving as a critical benchmark for predicting reactions with acids.
The Metal Power Rankings: A Guide to the Hidden Battle of Atoms
1. Introduction: The Secret Battle All Around Us
Namaste and a very warm welcome to you. Today, we are pulling back the curtain on a hidden power struggle that is happening literally all around us—in the pipes beneath our feet, the gadgets in our pockets, and the very structures of our cities. This is the secret battle in the world of metals.
Have you ever wondered why a piece of Magnesium dropped into acid fizzes away like a soda, while a piece of Gold sits perfectly still, untouched? That fizzing sound isn’t just a chemical byproduct; it is a “victory cry.” It is the sound of Hydrogen gas making a run for it as a more powerful metal literally kicks it out of the acid.
To solve this mystery, we use a specialized “League Table” for metals—a ranking system that dictates who dominates and who remains passive.
Did You Know? The “League Table” of metals, known as the Reactivity Series, is the ultimate playbook. It tells us which metals are the heavy-hitting champions of the chemical world and which ones are the quiet, unreactive underdogs.
To understand how these rankings are formed, we must step away from what we see with our eyes and peer into the invisible “atomic character” of the metals themselves.
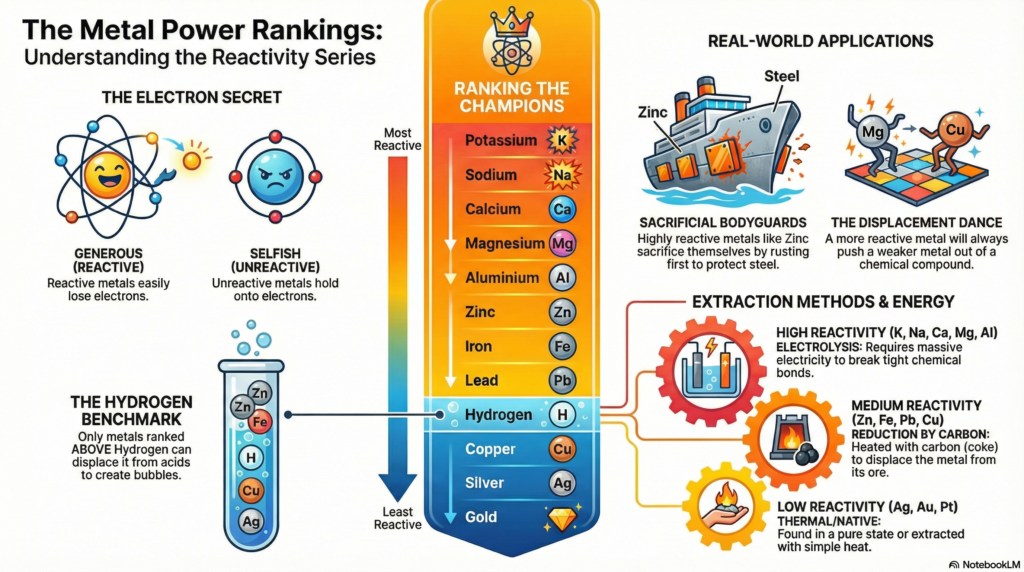
2. The Atomic Secret: Electron Generosity vs. Selfishness
At the heart of every chemical negotiation is a tiny particle: the electron. Reactivity is essentially a measure of an atom’s personality—specifically, its willingness to give its electrons away.
The Logic of Reactivity
| Highly Reactive Metals (The Champions) | Low Reactivity Metals (The Selfish) |
| Generous: These metals are “dying” to give their electrons away to others. | Selfish: These metals hold onto their electrons with an iron grip. |
| Active: They lose electrons easily to form positive ions (\text{M}^{n+}). | Passive: They prefer to stay in their pure, metallic state and rarely form ions. |
| Examples: Potassium (\text{K}), Sodium (\text{Na}) | Examples: Gold (\text{Au}), Silver (\text{Ag}) |
In chemistry, we define this “tendency to lose electrons” as a Redox reaction. It is the fundamental process of oxidation, expressed by the equation: \text{Metal} \longrightarrow \text{Metal}^{n+} + n\text{e}^-
This atomic generosity is the engine that drives the entire Reactivity Series. To map this hierarchy, scientists stepped into the role of chemical detectives, putting these metals through a series of rigorous trials.
——————————————————————————–
3. The Reactivity Series: Mapping the League Table
The Reactivity Series lists metals in descending order of their power. We use Hydrogen (\text{H}) as our crucial benchmark: metals above it can displace Hydrogen from acids, while those below it lack the strength to do so.
- Potassium (\text{K}) — The Reigning Champion
- Sodium (\text{Na})
- Calcium (\text{Ca})
- Magnesium (\text{Mg})
- Aluminium (\text{Al})
- Zinc (\text{Zn})
- Iron (\text{Fe})
- Lead (\text{Pb})
- (Hydrogen) (\text{H}) — The Reference Point
- Copper (\text{Cu})
- Silver (\text{Ag})
- Gold (\text{Au}) — The Least Reactive
The Memory Masterkey
To help this “League Table” stick in your mind, we use a brilliant Hindi mnemonic. Each word’s first sound provides a clue to the metal’s name and symbol in order:
“Phir Se Chala Mein Ae Zindagi Iss Liye Hum Chain Se Gaye”
| Mnemonic Word | Target Metal | Chemical Symbol |
| Phir | Potassium | \text{K} |
| Se | Sodium | \text{Na} |
| Chala | Calcium | \text{Ca} |
| Mein | Magnesium | \text{Mg} |
| Ae | Aluminium | \text{Al} |
| Zindagi | Zinc | \text{Zn} |
| Iss | Iron | \text{Fe} |
| Liye | Lead | \text{Pb} |
| Hum | Hydrogen | \text{H} |
| Chain | Copper | \text{Cu} |
| Se | Silver | \text{Ag} |
| Gaye | Gold | \text{Au} |
——————————————————————————–
4. The Science of the Ranking: Three Levels of Proof
Scientists proved these rankings by observing how metals behave under pressure in three distinct trials:
- The Water Test:
- Champions (\text{K, Na, Ca}) react vigorously with cold water, with Potassium practically exploding upon contact.
- Moderate Metals (\text{Mg, Zn, Fe}) are more reserved; they won’t react with cold water but will react with hot water or steam to form metal oxides.
- The Acid Fizz Test: By timing the rate of effervescence (the speed of \text{H}_2 bubbles), scientists measure a metal’s strength.
- The Nitric Acid (\text{HNO}_3) Exception: Usually, Nitric Acid doesn’t produce \text{H}_2 because it is a powerful oxidizer that turns the gas into water (\text{H}_2\text{O}). However, Magnesium (\text{Mg}) and Manganese (\text{Mn}) are such strong reducing agents that they can counteract this oxidizing action and force the production of Hydrogen if the acid is very dilute.
- The Displacement Dance: This is the ultimate test of power. A stronger metal is placed into a solution containing a weaker metal’s compound to see if it can forcibly “kick out” the underdog.
——————————————————————————–
5. The Displacement Dance: A Chemical Takeover
The “Displacement Dance” is a dramatic demonstration of atomic dominance. When a more generous metal meets a solution of a less reactive, “selfish” metal, a total takeover occurs.
Consider the battle between Zinc and Copper Sulfate:
\text{Zn}(\text{s}) + \text{CuSO}_4(\text{aq}) \longrightarrow \text{ZnSO}_4(\text{aq}) + \text{Cu}(\text{s})
The “So What?”: Zinc is higher on the league table and far more generous with its electrons. When it meets the Copper ions in the solution, Zinc gives its electrons away and moves into the solution as \text{Zn}^{2+}. It forces the Copper to take those electrons and return to its solid, metallic form. Zinc doesn’t just react; it wins the right to be part of the compound, displacing the weaker Copper.
——————————————————————————–
6. From Ores to Ocean Liners: Why This Matters
This ranking dictates the engineering of our modern world, from how we extract treasure from the earth to how we protect our greatest structures.
Metal Extraction Methods
| Reactivity Level | Extraction Method | Principle | Example |
| High (\text{K, Na, Ca, Mg, Al}) | Electrolysis | The metal is too reactive for carbon. Brute force electricity breaks molten bonds. | Aluminium from Alumina (\text{Al}_2\text{O}_3) |
| Medium (\text{Zn, Fe, Pb, Cu}) | Reduction by Carbon | Heated with carbon (coke), which is more reactive and displaces the metal. | Iron from Iron Oxide (\text{Fe}_2\text{O}_3) |
| Low (\text{Ag, Au, Pt}) | Thermal Decomposition | These metals are so unreactive they are often found in their pure, “native” state. | Gold found as pure nuggets. |
Sacrificial Protection: The Atomic Bodyguard
We use the “generosity” of metals to protect infrastructure from its greatest enemy: rust. This is the beauty of Sacrificial Protection.
- Galvanization: Coating steel with Zinc. Because Zinc is more reactive, it acts as the anode, losing its electrons (corroding) first to protect the steel underneath, which acts as the cathode.
- Sacrificial Anodes: On giant ships, massive blocks of Zinc or Magnesium are bolted to the steel hull. These blocks slowly sacrifice themselves to the harsh salt water, corroding away over years so the ship’s hull remains perfectly intact.
——————————————————————————–
7. Summary of Insights
- Reactivity is Electron Generosity: The most reactive metals are “Champions” because they lose electrons most easily.
- The Hydrogen Benchmark: Only metals above Hydrogen in the series have the power to displace it from acids to create that tell-tale fizz.
- Strategic Engineering: The series tells us how to extract metals from ores and how to use “sacrificial” metals as bodyguards for our ships and buildings.
As you look around today—from a rusty gate on a fence to a gleaming skyscraper—remember that you are witnessing a silent, powerful competition. Chemistry isn’t just a subject in a lab; it is the invisible logic that builds and protects our world. Keep exploring, and you will see these atomic “dances” in every object you touch!
The Reactivity Series of Metals and Industrial Properties
Based on 2 sources
| Metal | Chemical Symbol | Reactivity Level | Reaction with Water | Reaction with Dilute Acids | Extraction Method | Common Industrial Uses | Electron Tendency (Inferred) | Source |
|---|---|---|---|---|---|---|---|---|
| Potassium | K | High | Reacts violently with cold water | Reacts violently | Electrolysis | Sacrificial protection (via blocks) | Highest tendency to lose electrons | [1, 2] |
| Sodium | Na | High | Reacts vigorously with cold water | Reacts vigorously | Electrolysis | Not in source | Very high tendency to lose electrons | [1, 2] |
| Calcium | Ca | High | Reacts with cold water | Reacts with dilute acids | Electrolysis | Not in source | High tendency to lose electrons | [1, 2] |
| Magnesium | Mg | High | Reacts slowly with cold water; reacts with steam | Reacts well; reacts with very dilute nitric acid | Electrolysis | Sacrificial anodes for ship hulls and pipelines | High tendency to lose electrons | [1, 2] |
| Aluminium | Al | High | Not in source | Reacts well (protective oxide layer can slow reaction) | Electrolysis (from molten alumina) | Not in source | High tendency to lose electrons | [1, 2] |
| Zinc | Zn | Medium | Reacts with steam or hot water | Reacts with acids; faster rate of effervescence | Reduction by carbon | Galvanisation; sacrificial protection for steel | Moderate tendency to lose electrons | [1, 2] |
| Iron | Fe | Medium | Reacts with steam or hot water | Reacts slowly | Reduction by carbon (in a blast furnace) | Building material (protected by galvanisation) | Moderate tendency to lose electrons | [1, 2] |
| Lead | Pb | Medium | Not in source | Reacts slowly | Reduction by carbon | Not in source | Low tendency to lose electrons | [1, 2] |
| Copper | Cu | Low | No reaction | No reaction | Reduction by carbon | Not in source | High tendency to hold onto electrons | [1, 2] |
| Silver | Ag | Low | No reaction | No reaction | Thermal decomposition; found naturally in free state | Not in source | High tendency to hold onto electrons | [1, 2] |
| Gold | Au | Low | No reaction | No reaction | Found naturally in free state (native) | Not in source | Highest tendency to hold onto electrons | [1, 2] |
[1] The_Metal_Power_Rankings.mp4
[2] What happens when metals react with acids – Google Docs.pdfNotebookLM can be inaccurate; please double-check its responses.
