Foundations of Physical Reality: A Primer on Measurement and the Seven Dimensions
In the study of physics, we don’t just observe the world; we attempt to decode it. Measurement is the essential bridge between the abstract world of mathematics and the physical reality we touch. By establishing rigorous standards, we transform fleeting observations into universal truths that remain consistent whether they are calculated on Earth or in a distant galaxy.
1. The Philosophy of Measurement: Defining our Standards
Measurement is the act of comparing a physical quantity against a specific, internationally accepted reference standard known as a unit. Without units, numbers are orphaned; they have no context and no power to describe reality. Every physical measurement is the product of two inseparable components: the numerical value and the unit that gives it meaning.
Q = nu Where n is the numerical measure and u is the unit. Example: If a pendulum swings for 1.62 seconds, “1.62” is the numerical measure (n) and “second” is the unit (u).
The Architecture of Units
The universe presents a dizzying array of phenomena, but its underlying structure is surprisingly lean. We categorize units into two distinct tiers:
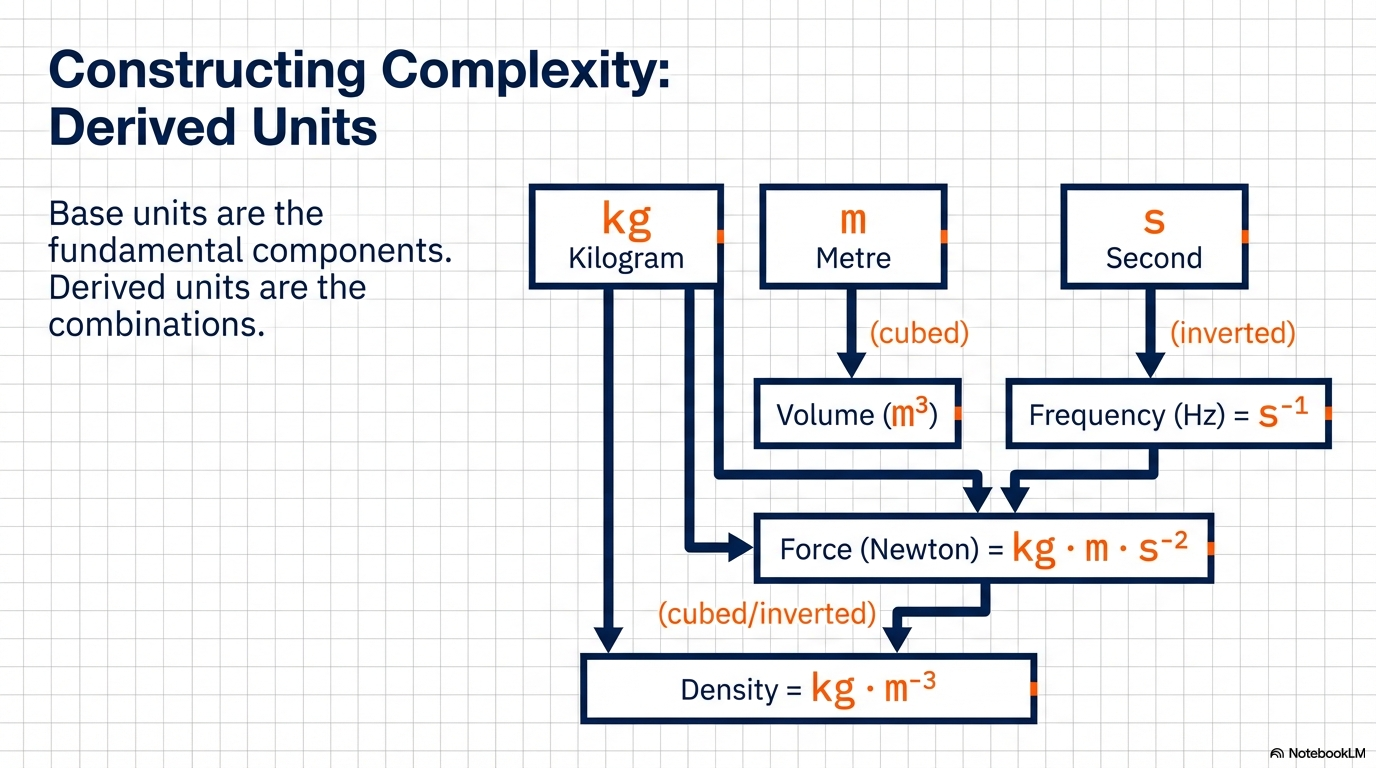
- Fundamental (Base) Units: These are the seven independent building blocks of the physical world. They cannot be simplified further or derived from one another.
- Derived Units: These are the “biological descendants” of the base units, formed by mathematical combinations (e.g., Velocity is Length divided by Time).
The “So What?”: This hierarchy is the foundation of scientific efficiency. We do not need to invent a unique, independent unit for every force or energy in the cosmos; we simply need seven fundamental pillars to describe the entire infinite landscape of physical quantities.
By establishing these pillars, we moved away from regional confusion toward a unified global language of science.
——————————————————————————–
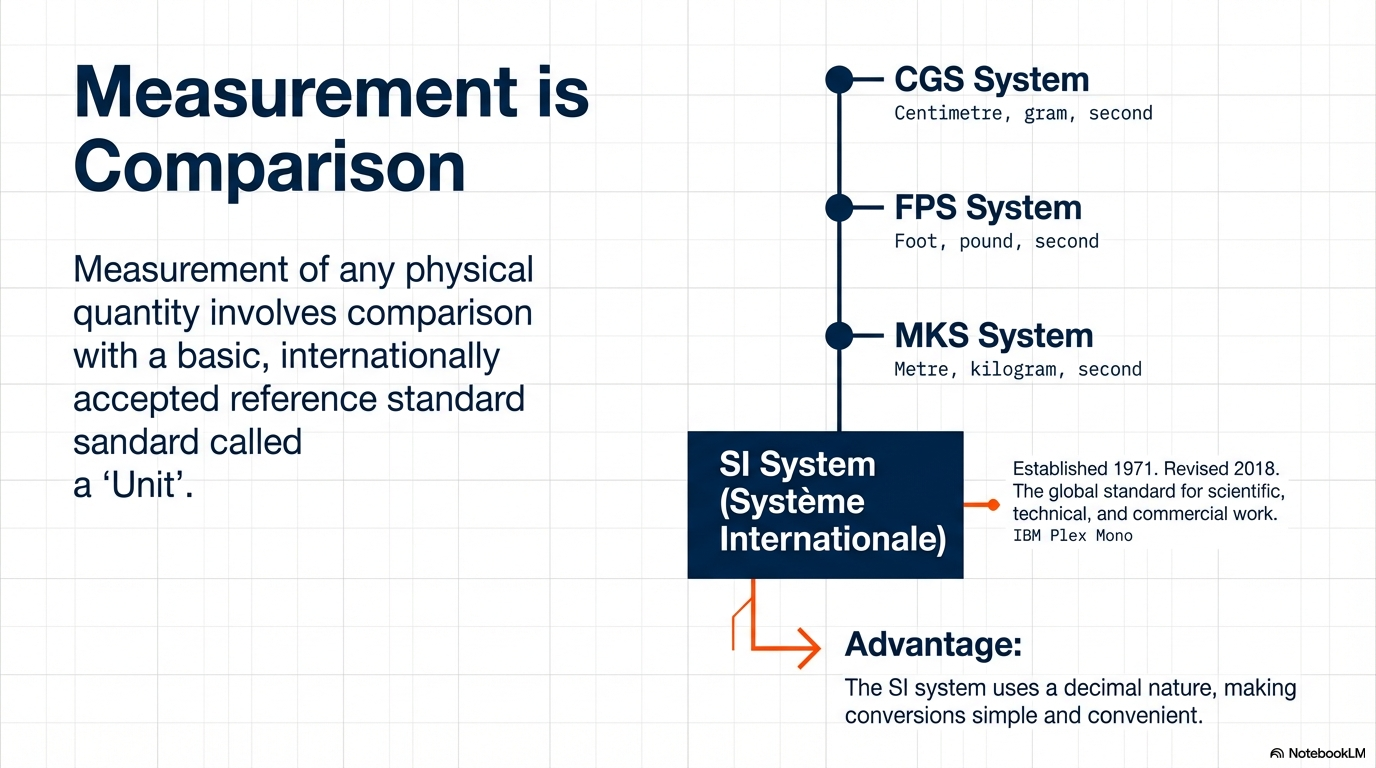
2. Evolution of the Global Standard: From MKS to SI
Historically, scientists were divided by their local systems of measurement. This lack of uniformity was a friction point for progress, leading to the development and eventual dominance of a single, standardized system.
| System | Length Unit | Mass Unit | Time Unit |
| CGS | Centimetre | Gram | Second |
| FPS (British) | Foot | Pound | Second |
| MKS | Metre | Kilogram | Second |
| SI (Modern) | Metre | Kilogram | Second |
The Perfection of the SI System
The Système Internationale d’ Unites (SI) is the modern evolution of the MKS system. Its primary advantage is its decimal-based structure. By using powers of ten, it eliminates the “human error” inherent in complex conversion factors—like the 5,280 feet in a mile—allowing the focus to remain entirely on the physics.
Beyond the base units, the SI system includes two “supplementary” dimensionless units to handle the geometry of our three-dimensional world:
- Radian (rad): Measures plane angles.
- Steradian (sr): Measures solid (3D) angles.
While these seven pillars provide the structure of our system, they are defined by the very constants of the universe itself.
——————————————————————————–
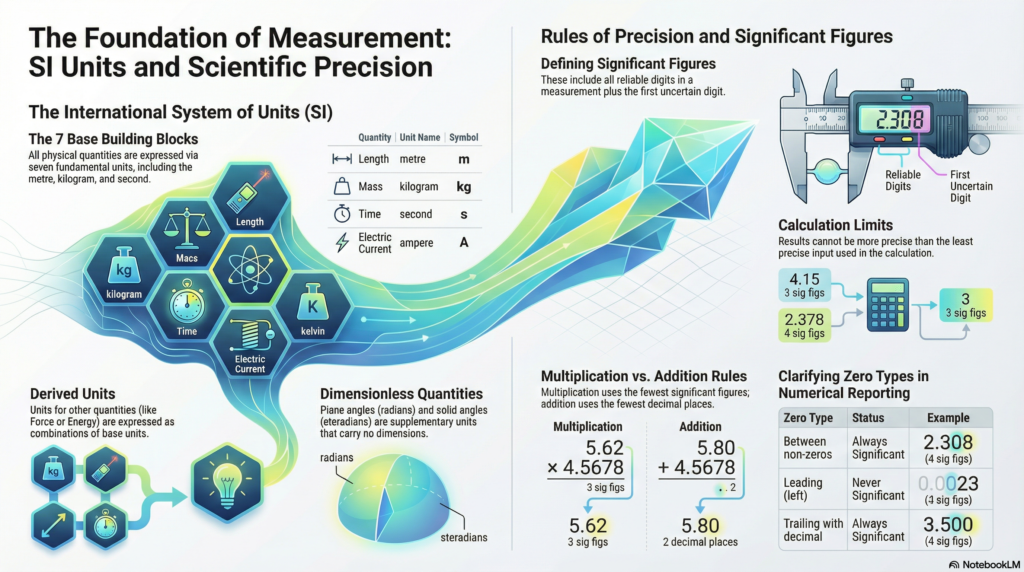
3. The Seven Pillars: The SI Base Quantities
As of the 2018 General Conference on Weights and Measures, the definitions of our base units have been anchored to fixed numerical values of universal constants. This ensures our measurements are no longer tied to physical artifacts that can degrade, but to the immutable laws of nature.
| Quantity | Unit Name | Symbol | Simplified Definition (Based on Constants) |
| Length | metre | m | Fixed to the speed of light (c = 299,792,458 m/s). |
| Mass | kilogram | kg | Defined by the Planck constant (h). |
| Time | second | s | Based on the frequency of the hyperfine transition of the caesium-133 atom. |
| Electric Current | ampere | A | Defined by the elementary charge (e). |
| Thermodynamic Temp. | kelvin | K | Defined by the Boltzmann constant (k). |
| Amount of Substance | mole | mol | Fixed to the Avogadro constant (N_A). Note: Elementary entities (atoms, ions, etc.) must be specified. |
| Luminous Intensity | candela | cd | Defined by the efficacy of monochromatic radiation. |
Pro-Tip: Precision is a Moving Target These definitions are revised periodically. As technology improves our ability to measure the “unperturbed” state of an atom or the speed of light, we update our definitions to ensure our scientific foundation remains the most precise tool in existence.
Even though our fundamental constants are perfect, the tools we use to measure them are not, which brings us to the necessity of reporting data with integrity.
——————————————————————————–
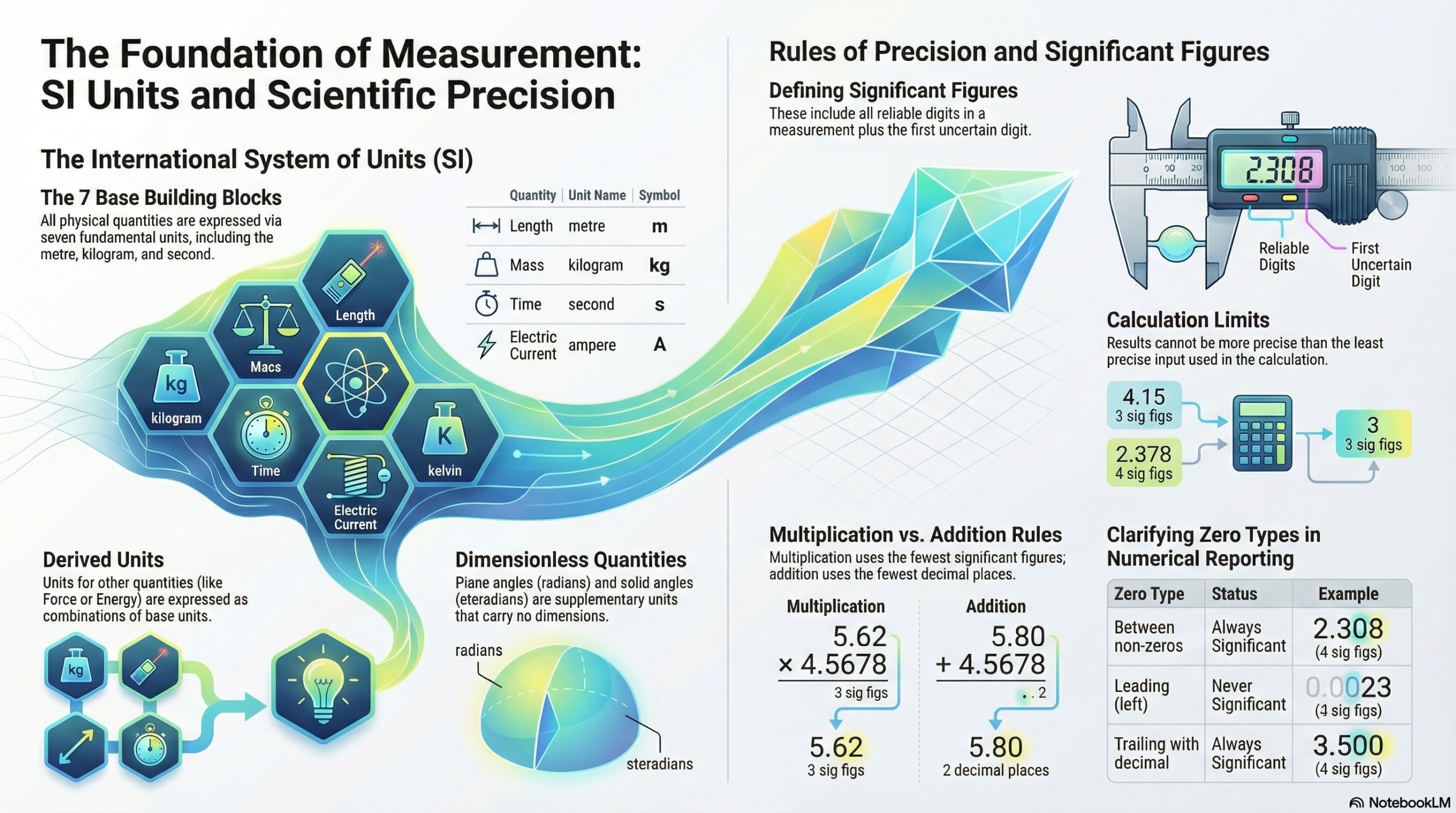
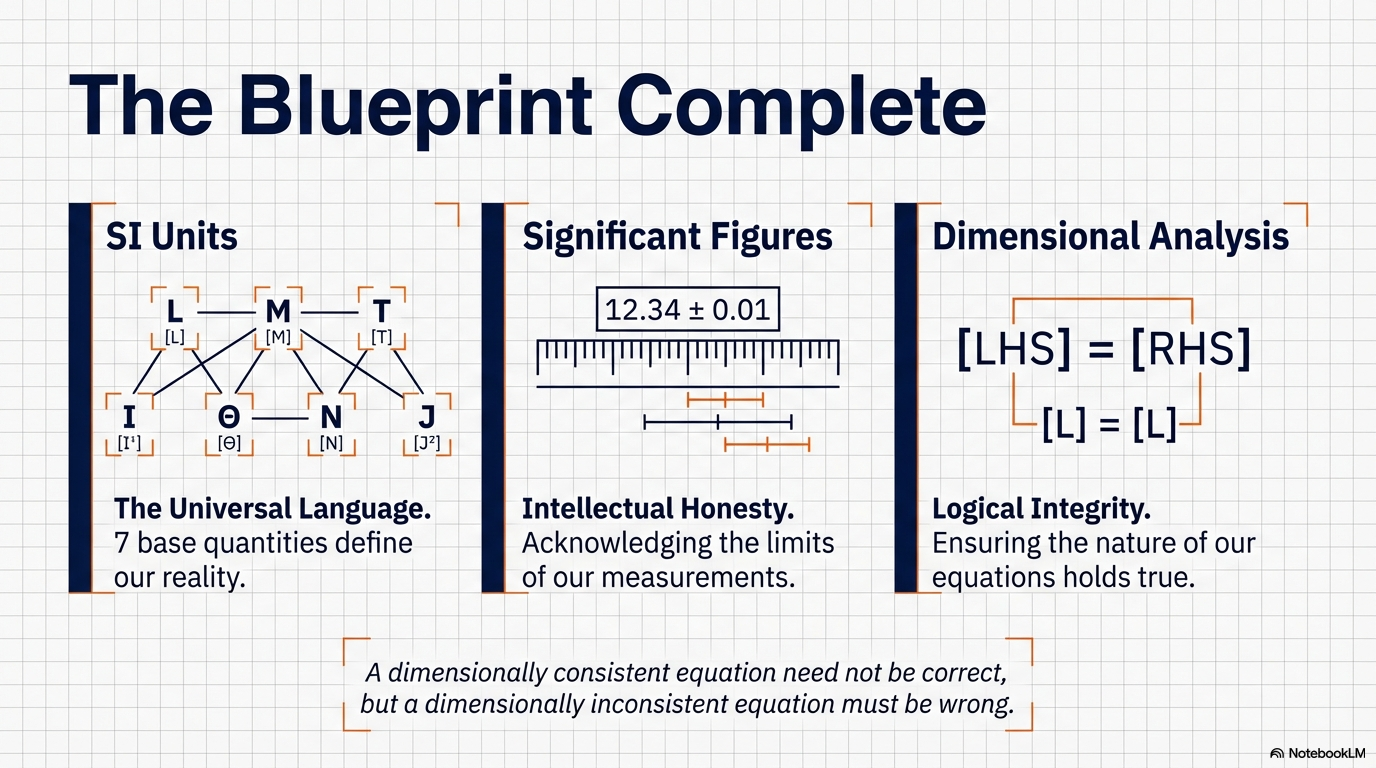
4. Precision and Integrity: Navigating Significant Figures
In physics, a number is a statement of confidence. Significant figures are not merely math rules; they reflect the precision of your measurement. A reported value includes all “reliable digits” plus the first “uncertain digit.” To report more is to be misleading; to report less is to lose data.
The Sig-Fig “Cheatsheet”
- Non-Zero Digits: Always significant. (287.5 has 4 sig figs.)
- The Sandwich Rule: Zeros between non-zero digits are significant. (2.308 has 4 sig figs.)
- The Leading Zero Rule: Zeros to the left of the first non-zero are never significant; they are just placeholders. (0.0023 has 2 sig figs.)
- The Trailing Zero Rule: Zeros at the end of a number are significant only if there is a decimal point. (4.700 has 4 sig figs; 12300 has only 3.)
Pro-Tip: The Scientific Notation Solution To remove all ambiguity regarding trailing zeros, always use Scientific Notation (a \times 10^b). In this format, every digit in the base number (a) is significant. For example, 4.700 \times 10^3 mm clearly has 4 significant figures, whereas 4700 mm is ambiguous.
The Arithmetic of Uncertainty
Your final result can never be more precise than your least precise input.
| Operation | Rule |
| Multiplication & Division | Result must match the total number of significant figures of the least precise input. |
| Addition & Subtraction | Result must match the number of decimal places of the least precise input. |
The Even-Odd Rounding Rule
When rounding a number where the insignificant digit to be dropped is exactly 5:
- If the preceding digit is even, leave it unchanged. (e.g., 2.745 becomes 2.74)
- If the preceding digit is odd, raise it by 1. (e.g., 2.735 becomes 2.74)
While significant figures protect the integrity of our numbers, dimensions protect the logic of our equations.
——————————————————————————–
5. The Seven Dimensions: The “DNA” of Physics
If a unit is the “suit of clothes” a number wears, the dimension is its biological species. Dimensions describe the qualitative nature of a physical quantity—what it is rather than how much of it there is. We denote these using square brackets:
- Length: [L]
- Mass: [M]
- Time: [T]
- Current: [A]
- Temperature: [K]
- Luminous Intensity: [cd]
- Amount of Substance: [mol]
Derived Dimensions: The Dimensional Fingerprint
Every complex quantity can be stripped down to its base dimensions. This “fingerprint” allows us to see the relationship between seemingly different concepts.
Example: Deriving Force
- Formula: \text{Force} = \text{mass} \times \text{acceleration}
- Acceleration is \text{velocity} / \text{time} = ([L]/[T]) / [T] = [LT^{-2}]
- Combine: [M] \times [LT^{-2}] = [MLT^{-2}]
This dimensional fingerprint is the ultimate “truth-test” for any physical law.
——————————————————————————–
6. Dimensional Analysis: The Scientist’s Error-Checking Tool
The Principle of Homogeneity is the “First Filter” of physics. It states that you can only add or subtract quantities that share the exact same dimensions. You cannot add a velocity to a force any more than you can add apples to ideas.
Case Study: Verifying Kinetic Energy (K)
If you are unsure of the formula for Kinetic Energy, dimensional analysis acts as a gatekeeper:
- Test K = ma: Dimensions are [M] \times [LT^{-2}] = [MLT^{-2}]. These are the dimensions of Force. (Ruled out)
- Test K = \frac{1}{2}mv^2: Dimensions are [M] \times [LT^{-1}]^2 = [ML^2T^{-2}]. This matches the dimensions of Energy/Work. (Dimensionally consistent)
The Logical Limits
Dimensional analysis is a powerful tool for (1) checking consistency and (2) deducing relations, but it has two vital limitations:
- It cannot identify dimensionless constants (like the 1/2 or 3/16 in energy formulas).
- It cannot distinguish between different quantities with the same dimensions (such as Work and Torque, which share the same “fingerprint”).
——————————————————————————–
7. Summary of Insights
Mastery Checklist
- Units as Standards: Every measurement is a comparison (nu) against an internationally accepted reference.
- The SI Advantage: Use the decimal-based SI system to minimize conversion errors.
- The Seven Pillars: Remember that the 2018 revisions anchored units to universal constants like the speed of light and the Caesium-133 frequency.
- Precision Integrity: Use Scientific Notation to resolve trailing zero ambiguity and apply the Even-Odd rule for rounding.
- Dimensional DNA: Treat dimensions ([M], [L], [T]) as the underlying structural logic of an equation.
- Homogeneity: Only add or subtract quantities with identical dimensions.
Mastering these foundations is your first step into the deep mysteries of the universe. By respecting the “DNA” of your measurements, you ensure that every calculation you perform is built on a solid, logical, and universal ground.
| Base quantity | Name | Symbol | Definition | Physical Dimensions | Source |
|---|---|---|---|---|---|
| Length | metre | m | The metre, symbol m, is the SI unit of length. It is defined by taking the fixed numerical value of the speed of light in vacuum c to be 299792458 when expressed in the unit ms−1, where the second is defined in terms of the caesium frequency ΔνCs. | [L] | [1] |
| Mass | kilogram | kg | The kilogram, symbol kg, is the SI unit of mass. It is defined by taking the fixed numerical value of the Planck constant h to be 6.62607015×10−34 when expressed in the unit Js, which is equal to kgm2s−1, where the metre and the second are defined in terms of c and ΔνCs. | [M] | [1] |
| Time | second | s | The second, symbol s, is the SI unit of time. It is defined by taking the fixed numerical value of the caesium frequency ΔνCs, the unperturbed ground-state hyperfine transition frequency of the caesium-133 atom, to be 9192631770 when expressed in the unit Hz, which is equal to s−1. | [T] | [1] |
| Electric current | ampere | A | The ampere, symbol A, is the SI unit of electric current. It is defined by taking the fixed numerical value of the elementary charge e to be 1.602176634×10−19 when expressed in the unit C, which is equal to As, where the second is defined in terms of ΔνCs. | [I] | [1] |
| Thermodynamic temperature | kelvin | K | The kelvin, symbol K, is the SI unit of thermodynamic temperature. It is defined by taking the fixed numerical value of the Boltzmann constant k to be 1.380649×10−23 when expressed in the unit JK−1, which is equal to kgm2s−2K−1, where the kilogram, metre and second are defined in terms of h, c and ΔνCs. | [Θ] | [1] |
| Amount of substance | mole | mol | The mole, symbol mol, is the SI unit of amount of substance. One mole contains exactly 6.02214076×1023 elementary entities. This number is the fixed numerical value of the Avogadro constant, NA, when expressed in the unit mol−1 and is called the Avogadro number. | [N] | [1] |
| Luminous intensity | candela | cd | The candela, symbol cd, is the SI unit of luminous intensity in a given direction. It is defined by taking the fixed numerical value of the luminous efficacy of monochromatic radiation of frequency 540×1012Hz, Kcd, to be 683 when expressed in the unit lmW−1, which is equal to cdsrW−1, or cdsrkg−1m−2s3. | [J] | [1] |